Immobilization of gold nanoparticles on folate-conjugated dendritic mesoporous silica-coated reduced graphene oxide nanosheets: a new nanoplatform for curcumin pH-controlled and targeted delivery
Abstract
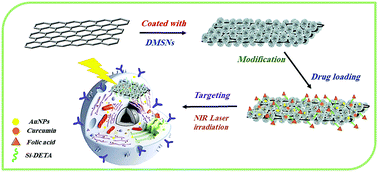
In the present study, a new sandwich-like nanocomposite as a multifunctional smart nanocarrier for curcumin (Cur) targeted delivery and cell imaging was prepared by immobilization of gold nanoparticles on folic acid-modified dendritic mesoporous silica-coated reduced graphene oxide nanosheets (AuNPs@GFMS). The physical and chemical properties of the nanocomposite were investigated by atomic force microscopy (AFM), transmission electron microscopy (TEM), X-ray diffraction (XRD), UV-Vis, field-emission scanning electron microscopy (FE-SEM), Fourier transformation infrared (FT-IR), and Brunauer–Emmett–Teller (BET) surface area analysis. The nanocarrier exhibits a number of interesting properties, including good biocompatibility, biodegradability, and suitable surface area, which results in high drug loading capacity. In addition, this new drug delivery system showed sustained-release and pH-responsive properties. The in vitro cytotoxicity test of the free curcumin, free nanocarrier (AuNPs@GFMS), curcumin-loaded folate-conjugated nanocarriers (Cur-AuNPs@GFMS), and curcumin-loaded nanocarriers without folate-conjugation (Cur-AuNPs@GAMS) against two human cancer cell lines, including MCF-7 (human breast carcinoma cell lines) and A549 (human lung carcinoma cell lines) demonstrated that the therapeutic efficacy of Cur-AuNPs@GFMS is significantly greater than those of other compounds because the cancerous cells can uptake the folate-conjugated drug nanocarrier via a receptor-mediated mechanism. Fluorescence microscopic images and different staining techniques were also used to visualize the cellular uptake, anticancer activity, specific targeting ability, and photothermal potency of Cur-AuNPs@GFMS toward the MCF-7 cancer cells. The obtained results proved that the proposed system, Cur-AuNPs@GFMS, can be used as a potent anticancer agent in targeted cancer therapies for breast cancer.


 Please wait while we load your content...
Please wait while we load your content...