No unjamming transition in a Voronoi model of biological tissue
Abstract
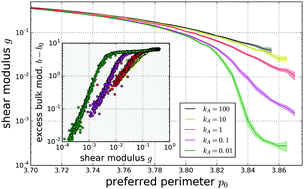
Vertex models are a popular approach to simulating the mechanical and dynamical properties of dense biological tissues, describing the tissue as a network of polygons. Recently a class of two-dimensional vertex models was shown to exhibit a disordered rigidity transition controlled by the preferred cellular geometry, which was subsequently echoed by experimental findings. An attractive variant of these models uses a Voronoi tessellation to describe the cells, which reduces the number of degrees of freedom as compared the original vertex model. The Voronoi model was also endowed with a non-equilibrium model of cellular motility, leading to rich, glassy behavior. This glassy behavior was suggested to be inextricably linked to an underlying jamming transition. We test this conjecture, exploring the low-effective-temperature limit of the 2D Voronoi model by studying cell trajectories from detailed dynamical simulations in combination with rigidity measurements of energy-minimized disordered cell configurations. We find that the zero-temperature limit of this model has no unjamming transition. We show that this absence of an unjamming transition is intimately linked to the marginality of the model, i.e. the fact that the constraints imposed on cell areas and perimeters precisely balance the number of degrees of freedom in the model. Our work suggests that constraint counting arguments are useful to understand rigidity in a broad class of models of dense biological tissues.


 Please wait while we load your content...
Please wait while we load your content...