A strategically managed rechargeable battery system with a neutral methyl viologen anolyte and an acidic air-cathode enabled by a mediator-ion solid electrolyte†
Abstract
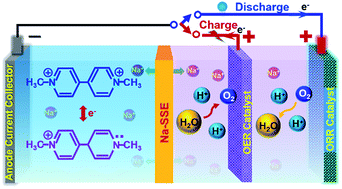
Redox flow batteries with organic electrode materials are attracting much attention. Previous research efforts have been focusing on liquid-phase electrodes on both the anode and cathode sides. Since batteries based on air cathodes can provide immense advantages, coupling a liquid organic electrode with a gaseous air cathode could offer multiple benefits in terms of cost, safety, and energy density. Herein we present a liquid–gaseous battery system with an aqueous methyl viologen (MV) anode and an air cathode. However, under the traditional battery operation principle with the same electrolyte at the anode and cathode, the resulting MV–air battery will not be able to provide a reasonable voltage for practical applications. In this study, the cell voltage of the MV–air chemistry is strategically manipulated by using an acidic cathode electrolyte (catholyte) and a neutral anode electrolyte (anolyte). To operate a battery with different electrolytes at the anode and cathode, a sodium-ion (Na+-ion) conductive solid-state electrolyte (Na-SSE) membrane is employed to physically and electrically separate the two electrodes. The shuttling of sodium ions via the Na-SSE balances the ionic charge transfer between the two electrodes and sustains the redox reactions at the air cathode and the MV anode.
- This article is part of the themed collection: 2018 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...