Uncovering the mechanisms of electrolyte permeation in porous electrodes for redox flow batteries through real time in situ 3D imaging
Abstract
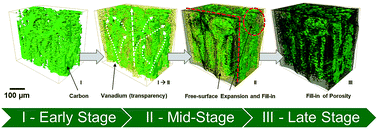
Increasing energy demands have expedited the need for grid-scale energy storage solutions. High power densities have been achieved using carbon-based electrodes in all-vanadium redox flow batteries. However, fundamental limitations must be overcome to improve viability. This study addresses the infiltration of vanadium solutions into dry electrodes using time-resolved 3D X-ray tomography, thus relating the rate of permeation and other wetting phenomena to microstructural characteristics and surface properties of carbon fibers. It is shown that electrolyte infiltration proceeds according to a non-uniform progression front, with small channel-like pores filled in first and remaining larger pores filled only after 6 h. The vanadium concentration affects the infiltration rate, being faster for 0.5 M VOSO4 while slower for both higher and lower concentrations. The infiltration mechanism is strongly correlated with the temporal decrease in the contact angle, revealing that fibers become more hydrophilic as they are treated by the electrolyte solution. Modification of carbon surface groups through plasma treatment boosts the infiltration process. The results reveal counter-intuitive behaviors of the electrolyte flow whereby the capillary driven flow is found to be secondary to the primary wetting mechanisms. Uncovering these physical phenomena is essential for operational procedures in flow batteries and avoiding cell degradation.
- This article is part of the themed collection: 2018 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...