A hybrid molecular photoanode for efficient light-induced water oxidation†
Abstract
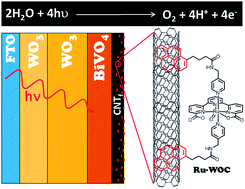
A hybrid photoanode comprising a multilayered heterostructured WO3/BiVO4 semiconductor and a molecular water oxidation catalyst Ru(tda)(py-pyr)2 (Ru-WOC, where tda is [2,2′:6′,2′′-terpyridine]-6,6′′-dicarboxylato and py-pyr is 4-(pyren-1-yl)-N-(pyridin-4-ylmethyl)butanamide) is described. Both elements are linked by a highly conductive carbon nanotube fibre film (CNTf), which acts as charge transfer and anchoring platform, to which the catalyst is attached through π–π stacking interactions. Photoelectrochemical characterization of the resulting electrodes shows that the full photoanode WO3/BiVO4/CNTf/Ru-WOC outperforms the bare WO3/BiVO4 electrode in the potential range 0.3–0.8 V vs. NHE at pH 7, with current densities enhanced by 0.05–0.29 mA cm−2. Bulk electrolysis experiments and oxygen gas measurements show that the enhanced photocurrent is due to the catalytic water oxidation reaction. Detailed electrochemical impedance spectroscopy (EIS) analysis is used to investigate the roles of the multiple layers involved in the process. The CNTf/Ru–WOC interface is responsible for increasing charge accumulation and reducing recombination phenomena. The CNTf is able to hold the charge produced from light absorbed by the WO3/BiVO4 semiconductor, as shown by the high capacitive values observed for a WO3/BiVO4/CNTf electrode in the whole range of studied potentials (0.15–0.85 V vs. NHE). Furthermore, Ru-WOC transfers the charge to the solution through fast water oxidation catalysis. This is supported by the low resistivity shown by the full WO3/BiVO4/CNTf/Ru-WOC electrode at low potentials (E < 0.5 V vs. NHE). The robustness and high catalytic rate of Ru-WOC ensures the proper performance of the hybrid photoelectrode device. The latter is particularly important, as it provides opportunities to improve the performance of photoanodes for the water oxidation reaction based on the easy modification of ligands in the molecular catalyst to tune its structural, electronic, and catalytic properties. This is a unique advantage compared with commonly used catalysts based on metal oxides or oxy(hydroxides), which have limited tunability.
- This article is part of the themed collection: Artificial Photosynthesis - From Sunlight to Fuels and Valuable Products for a Sustainable Future


 Please wait while we load your content...
Please wait while we load your content...