An ionic liquid based sodium metal-hybrid supercapacitor-battery†
Abstract
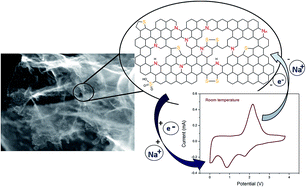
There is growing interest in developing sodium based energy storage devices as alternatives to Li for large-scale energy storage. We report a highly stable hybrid system comprising a sodium metal anode, a highly porous N/S co-doped mesoporous carbon cathode and the non-flammable ionic liquid electrolyte N-propyl-N-methyl pyrrolidinium bis(fluorosulfonyl) imide (C3mpyrFSI). This hybrid device operates at 100% coulombic efficiency and shows almost complete capacity retention over 3000 cycles. Owing to the reversible cathode reactions between Na+ and N/S functionalities on the carbon surface, a very high capacity of 716 mA h g−1 (at a rate of C/16) was achieved between 3.8 V and 0.005 V vs. Na+/Na. An optimised device could provide energy density as high as 263 W h kg−1. At high rate, the devices achieved power density of 1463 W kg−1. These metrics increase to 270 W h kg−1 and 3822 W kg−1 at 50 °C, highlighting the excellent combination of a supercapacitor-type cathode with a sodium metal anode and an ionic liquid electrolyte for large-scale energy storage.


 Please wait while we load your content...
Please wait while we load your content...