Electric-field induced bistability in single-molecule conductance measurements for boron coordinated curcuminoid compounds†
Abstract
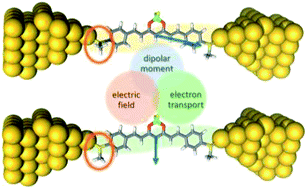
We have studied the single-molecule conductance of a family of curcuminoid molecules (CCMs) using the mechanically controlled break junction (MCBJ) technique. The CCMs under study contain methylthio (MeS–) as anchoring groups: MeS-CCM (1), the free-ligand organic molecule, and two coordination compounds, MeS-CCM-BF2 (2) and MeS-CCM-Cu (3), where ligand 1 coordinates to a boron center (BF2 group) and to a CuII moiety, respectively. We found that the three molecules present stable molecular junctions allowing detailed statistical analysis of their electronic properties. Compound 3 shows a slight increase in the conductance with respect to free ligand 1, whereas incorporation of BF2 (compound 2) promotes the presence of two conductance states in the measurements. Additional experiments with control molecules point out that this bistability is related to the combination of MeS– anchoring groups and the BF2 moiety within the structure of the molecules. Theoretical calculations show that this can be explained by the presence of two conformers once compound 2 is anchored between the gold electrodes. An energy minimum is found for a flat structure but there is a dramatic change in the magnitude and orientation of dipole moment (favouring a non-flat conformer in the presence of an external electric field) due to a conformational change of one of the terminal MeS– groups. The results thus point to an intricate interplay between the applied bias voltage and the molecule dipole moment which could be the basis for designing new molecules aiming at controlling their conformation in devices.


 Please wait while we load your content...
Please wait while we load your content...