Design of efficient bifunctional catalysts for direct conversion of syngas into lower olefins via methanol/dimethyl ether intermediates†
Abstract
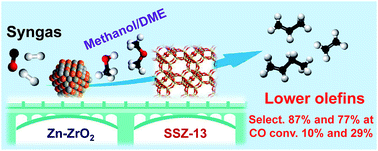
The direct conversion of syngas into lower olefins is a highly attractive route for the synthesis of lower olefins. The selectivity of lower olefins via the conventional Fischer–Tropsch (FT) synthesis is restricted to ∼60% with high CH4 selectivity due to the limitation by the Anderson–Schulz–Flory (ASF) distribution. Here, we report the design of bifunctional catalysts for the direct conversion of syngas into lower olefins with selectivity significantly breaking the ASF distribution. The selectivity of C2–C4 olefins reached 87% at a CO conversion of 10% and was sustained at 77% by increasing CO conversion to 29% over a bifunctional catalyst composed of Zn-doped ZrO2 nanoparticles and zeolite SSZ-13 nanocrystals. The selectivity of CH4 was lower than 3% at the same time. It is demonstrated that the molar ratio of Zn/Zr, the density of Brønsted acid sites of SSZ-13 and the proximity of the two components play crucial roles in determining CO conversion and lower-olefin selectivity. Our kinetic studies indicate that methanol and dimethyl ether (DME) are key reaction intermediates, and the conversion of syngas to methanol/DME is the rate-determining step over the bifunctional catalyst. Formate and methoxide species have been observed on Zn-doped ZrO2 surfaces during the activation of CO in H2, and the formed methanol/DME are transformed into lower olefins in SSZ-13.
- This article is part of the themed collections: Celebrating a Century of Excellency in Chemistry at Xiamen University and Editor’s Choice – Toshiharu Teranishi


 Please wait while we load your content...
Please wait while we load your content...