Highly stable and porous porphyrin-based zirconium and hafnium phosphonates – electron crystallography as an important tool for structure elucidation†
Abstract
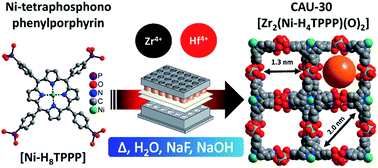
The Ni-metallated porphyrin-based tetraphosphonic acid (Ni-tetra(4-phosphonophenyl)porphyrin, Ni-H8TPPP) was used for the synthesis of highly porous metal phosphonates containing the tetravalent cations Zr4+ and Hf4+. The compounds were thoroughly characterized regarding their sorption properties towards N2 and H2O as well as thermal and chemical stability. During the synthesis optimization the reaction time could be substantially decreased under stirring from 24 to 3 h in glass vials. M-CAU-30, [M2(Ni-H2TPPP)(OH/F)2]·H2O (M = Zr, Hf) shows exceptionally high specific surface areas for metal phosphonates of aBET = 1070 and 1030 m2 g−1 for Zr- and Hf-CAU-30, respectively, which are very close/correspond to the theoretical values of 1180 and 1030 m2 g−1. CAU-30 is always obtained as mixtures with one mol ZrO2/HfO2 per formula unit as proven by TEM, electron diffraction, TG and elemental analysis. Hence experimentally derived specific surface areas are 970 and 910 m2 g−1, respectively. M-CAU-30 is chemically stable in the pH range 0 to 12 in HCl/NaOH and thermally up to 420 °C in air as determined by variable-temperature powder X-ray diffraction (VT-PXRD). The crystal structure of M-CAU-30 was determined by combining electron diffraction tomography for structure solution and powder X-ray diffraction data for the structure refinement. The crystal structure consists of chains of corner sharing MO6 octahedra interconnected by the partly deprotonated linker molecules Ni-H2TPPP6−. Thus 1D channels with pore diameters of 1.3 × 2.0 nm are formed. The redox activity of Zr-CAU-30 was investigated by cyclic voltammetry resulting in a reversible redox process at a half-wave potential of E1/2 = −0.649 V.


 Please wait while we load your content...
Please wait while we load your content...