Detection and identification of designer drugs by nanoparticle-based NMR chemosensing†
Abstract
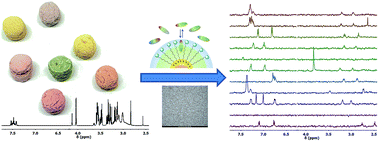
Properly designed monolayer-protected nanoparticles (2 nm core diameter) can be used as nanoreceptors for selective detection and identification of phenethylamine derivatives (designer drugs) in water. The molecular recognition mechanism is driven by the combination of electrostatic and hydrophobic interactions within the coating monolayer. Each nanoparticle can bind up to 30–40 analyte molecules. The affinity constants range from 105 to 106 M−1 and are modulated by the hydrophobicity of the aromatic moiety in the substrate. Detection of drug candidates (such as amphetamines and methamphetamines) is performed by using magnetization (NOE) or saturation (STD) transfer NMR experiments. In this way, the NMR spectrum of the drug is isolated from that of the mixture, allowing broad-class multianalyte detection and even identification of unknowns. The introduction of a dimethylsilane moiety in the coating monolayer allows performing STD experiments in complex mixtures. In this way, a detection limit of 30 μM is reached with standard instruments.
- This article is part of the themed collection: 2018 ChemSci Pick of the Week Collection


 Please wait while we load your content...
Please wait while we load your content...