A chiral nickel DBFOX complex as a bifunctional catalyst for visible-light-promoted asymmetric photoredox reactions†
Abstract
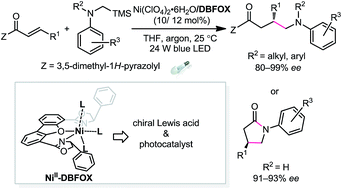
The enantioselective photoredox reaction of α,β-unsaturated carbonyl compounds and tertiary/secondary α-silylamines was enabled by a readily available single NiII–DBFOX catalyst (DBFOX = 4,6-bis((R)-4-phenyl-4,5-dihydrooxazol-2-yl)dibenzo[b,d]furan) under visible light conditions. The non-precious chiral catalyst is involved in the photochemical process to initiate single electron transfer and at the same time provides a well-organized chiral environment for the subsequent radical transformations. Good to excellent enantioselectivities (80–99% ee) were obtained for the formation of chiral γ-amino carboxylic acid derivatives and γ-lactams.
- This article is part of the themed collection: Celebrating a Century of Excellency in Chemistry at Xiamen University


 Please wait while we load your content...
Please wait while we load your content...