A thioether-directed palladium-cleavable linker for targeted bioorthogonal drug decaging†
Abstract
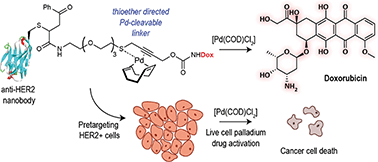
We describe the development of a bifunctional linker that simultaneously allows site-specific protein modification and palladium-mediated bioorthogonal decaging. This was enabled by a thioether binding motif in the propargyl carbamate linker and a readily available palladium complex. We demonstrate the efficiency of this reaction by controlled drug release from a PEGylated doxorubicin prodrug in cancer cells. The linker can be easily installed into cysteine bearing proteins which we demonstrated for the construction of an anti-HER2 nanobody–drug conjugate. Targeted delivery of the nanobody drug conjugate showed effective cell killing in HER2+ cells upon palladium-mediated decaging.
- This article is part of the themed collections: Most popular 2018-2019 chemical biology articles and 2019 International Open Access Week Collection


 Please wait while we load your content...
Please wait while we load your content...