ROS scavenging Mn3O4 nanozymes for in vivo anti-inflammation†
Abstract
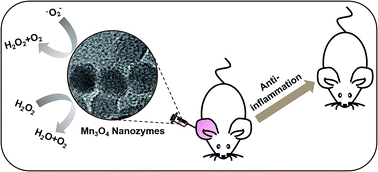
Reactive oxygen species (ROS)-induced oxidative stress is linked to various diseases, including cardiovascular disease and cancer. Though highly efficient natural ROS scavenging enzymes have been evolved, they are sensitive to environmental conditions and hard to mass-produce. Therefore, enormous efforts have been devoted to developing artificial enzymes with ROS scavenging activities. Among them, ROS scavenging nanozymes have recently attracted great interest owing to their enhanced stability, multi-functionality, and tunable activity. It has been implicated that Mn-contained nanozymes would possess efficient ROS scavenging activities, however only a few such nanozymes have been reported. To fill this gap, herein we demonstrated that Mn3O4 nanoparticles (NPs) possessed multiple enzyme mimicking activities (i.e., superoxide dismutase and catalase mimicking activities as well as hydroxyl radical scavenging activity). The Mn3O4 nanozymes therefore significantly scavenged superoxide radical as well as hydrogen peroxide and hydroxyl radical. Moreover, they were not only more stable than the corresponding natural enzymes but also superior to CeO2 nanozymes in terms of ROS elimination. We showed that the Mn3O4 NPs not only exhibited excellent ROS removal efficacy in vitro but also effectively protected live mice from ROS-induced ear-inflammation in vivo. These results indicated that Mn3O4 nanozymes are promising therapeutic nanomedicine for treating ROS-related diseases.
- This article is part of the themed collections: Celebrating 15 years of exceptional research in Chemical Science, Most popular 2018-2019 analytical chemistry articles, Most popular 2018-2019 nanoscience articles and Most Impactful Nanoscience Articles


 Please wait while we load your content...
Please wait while we load your content...