Incorporation of redox-inactive cations promotes iron catalyzed aerobic C–H oxidation at mild potentials†
Abstract
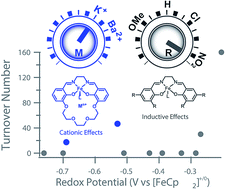
The synthesis and characterization of the Schiff base complexes Fe(II) (2M) and Fe(III)Cl (3M), where M is a K+ or Ba2+ ion incorporated into the ligand, are reported. The Fe(III/II) redox potentials are positively shifted by 440 mV (2K) and 640 mV (2Ba) compared to Fe(salen) (salen = N,N′-bis(salicylidene)ethylenediamine), and by 70 mV (3K) and 230 mV (3Ba) compared to Fe(Cl)(salen), which is likely due to an electrostatic effect (electric field) from the cation. The catalytic activity of 3M towards the aerobic oxidation of allylic C–H bonds was explored. Prior studies on iron salen complexes modified through conventional electron-donating or withdrawing substituents found that only the most oxidizing derivatives were competent catalysts. In contrast, the 3M complexes, which are significantly less oxidizing, are both active. Mechanistic studies comparing 3M to Fe(salen) derivatives indicate that the proximal cation contributes to the overall reactivity in the rate determining step. The cationic charge also inhibits oxidative deactivation through formation of the corresponding Fe2-μ-oxo complexes, which were isolated and characterized. This study demonstrates how non-redox active Lewis acidic cations in the secondary coordination sphere can be used to modify redox catalysts in order to operate at milder potentials with a minimal impact on the reactivity, an effect that was unattainable by tuning the catalyst through traditional substituent effects on the ligand.


 Please wait while we load your content...
Please wait while we load your content...