A mitochondria-targeted nanoradiosensitizer activating reactive oxygen species burst for enhanced radiation therapy†
Abstract
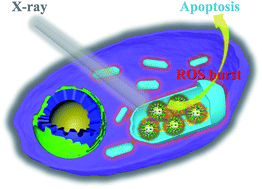
Radiation therapy (RT) has been widely used for malignant tumor treatment. However, the large dosage of ionizing radiation and high frequency of radiotherapy in clinical cancer therapy cause severe damage to normal tissues adjacent to tumors. Therefore, how to increase the local treatment efficacy and reduce the damage to normal tissues has been a challenge for RT. Herein, we developed a novel strategy for enhanced RT based on a mitochondria targeted titanium dioxide-gold nanoradiosensitizer. When irradiated with X-rays, the nanosensitizer could produce reactive oxygen species (ROS) in the mitochondria, which induced the domino effect on the ROS burst. The overproduced ROS accumulated in mitochondria, resulting in mitochondrial collapse and irreversible cell apoptosis. A colony formation assay indicated that the cell survival rate when incubated with the mitochondrial targeted nanosensitizer was significantly lower than that of non-targeted groups. As demonstrated by in vivo experiments, the tumor was significantly suppressed even just once RT with the nanosensitizer.
- This article is part of the themed collections: Most popular 2018-2019 chemical biology articles, In celebration of Chinese New Year and Most impactful Chemical Biology articles of 2018


 Please wait while we load your content...
Please wait while we load your content...