Organocatalytic controlled/living ring-opening polymerization of cyclotrisiloxanes initiated by water with strong organic base catalysts†
Abstract
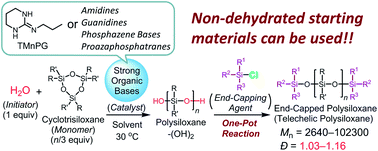
Organocatalytic controlled/living ring-opening polymerization of cyclotrisiloxanes, such as hexamethylcyclotrisiloxane, 1,3,5-trimethyl-1,3,5-triphenylcyclotrisiloxane, 1,3,5-trimethyl-1,3,5-trivinylcyclotrisiloxane, and 1,3,5-trimethyl-1,3,5-tris(3,3,3-trifluoropropyl)cyclotrisiloxane, using water as an initiator and strong organic bases, such as amidines, guanidines, phosphazene bases, and proazaphosphatrane, as catalysts produced a variety of polysiloxanes with controlled number-average molecular weights (Mn = 2.64–102.3 kg mol−1), narrow polydispersity (Đ = 1.03–1.16), and well-defined symmetric structures. Controlled syntheses of statistical copolymers and triblock copolymers were achieved by copolymerizations of two cyclotrisiloxanes. Various terminal functionalities were successfully introduced by the end-capping reaction of propagating polysiloxanes using functional chlorosilanes. Kinetic investigations demonstrated that the polymerization proceeded through the initiator/chain-end activation mechanism, namely activations of water in the initiation reaction and of terminal silanols in propagating polysiloxanes in the propagation reaction. Catalytic activities of strong organic bases were revealed to depend on their Brønsted basicity and efficiency of the proton transfer in the initiation and propagation reactions. Guanidines possessing an R–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(N)–NH–R′ unit, in particular 1,3-trimethylene-2-propylguanidine, showed excellent performance as a catalyst. In this system, even non-dehydrated solvents are usable for the polymerization.
C(N)–NH–R′ unit, in particular 1,3-trimethylene-2-propylguanidine, showed excellent performance as a catalyst. In this system, even non-dehydrated solvents are usable for the polymerization.


 Please wait while we load your content...
Please wait while we load your content...