The harpooning mechanism as evidenced in the oxidation reaction of the Al atom†
Abstract
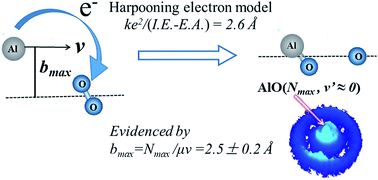
The harpooning mechanism has long been proposed for elementary reaction dynamics involving metals. It is characterized by an initial electron transfer (ET) process from the metal to the oxidant molecule. For the titled reaction Al + O2, the ET distance can be predicted to be 2.6 Å by simply calculating the energy difference between the ionization energy of the Al atom and the electron affinity of the O2 molecule. Hereby we experimentally derived the maximum impact parameter bmax of 2.5 ± 0.2 Å for the titled reaction, in consistency with the predicted ET distance. This derivation of bmax was achieved by using the crossed molecular beam experiment at a collision energy of 507 cm−1 (i.e. 1.45 kcal mol−1) with a high resolution time-sliced ion velocity imaging detection of the state-selective AlO products based on the (1 + 1) resonance-enhanced multiphoton ionization. The small rotational constant of the AlO(X2Σ+) radical (Be = 0.6413 cm−1) facilitated the formation of the AlO(v = 0) products in high rotational levels up to the energetically limited state, Nmax = 52, with an almost zero velocity mapping. Hence, in this extreme angular momentum disposal case, the collisional orbital angular momentum l was nearly completely channeled into the product rotational angular momentum as a consequence of the conservations of energy and angular momentum, offering a reaction system that breaks the restriction of kinematically favored mass combination in order to obtain information on the impact parameters. The present study yields the first direct derivation of bmax from the maximum rotational level of products under the experimental condition with the recoil energy E′T ≈ 0. This, in turn, provides solid evidence in supporting the harpooning mechanism.


 Please wait while we load your content...
Please wait while we load your content...