Stimuli-responsive thiol-epoxy networks with photo-switchable bulk and surface properties†
Abstract
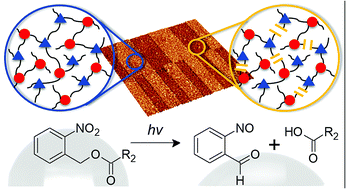
In the present work, the versatile nature of o-nitrobenzyl chemistry is used to alter bulk and surface properties of thiol-epoxy networks. By introducing an irreversibly photocleavable chromophore into the click network, material properties such as wettability, solubility and crosslink density are switched locally by light of a defined wavelength. The synthesis of photo-responsive thiol-epoxy networks follows a photobase-catalyzed nucleophilic ring opening of epoxy monomers with photolabile o-nitrobenzyl ester (o-NBE) groups across multi-functional thiols. To ensure temporal control of the curing reaction, a photolatent base is employed releasing a strong amidine-type base upon light exposure, which acts as an efficient catalyst for the thiol epoxy addition reaction. The spectral sensitivity of the photolatent base is extended to the visible light region by adding a selected photosensitizer to the resin formulation. Thus, in the case of photoactivation of the crosslinking reaction the photorelease of the base does not interfere with the absorbance of the o-NBE groups. Once the network has been formed, the susceptibility of the o-NBE groups towards photocleavage reactions is used for a well-defined network degradation upon UV exposure. Sol–gel analysis evidences the formation of soluble species, which is exploited to inscribe positive tone micropatterns by photolithography. Along with the localized tuning of network structure, the irreversible photoreaction is exploited to change the surface wettability of thiol-epoxy networks. The contact angle of water significantly decreases upon UV exposure due to the photo-induced formation of hydrophilic cleavage products enabling the inscription of domains with different surface wettability by photolithography.


 Please wait while we load your content...
Please wait while we load your content...