Luminescence characteristics of rare-earth-doped barium hexafluorogermanate BaGeF6 nanowires: fast subnanosecond decay time and high sensitivity in H2O2 detection†
Abstract
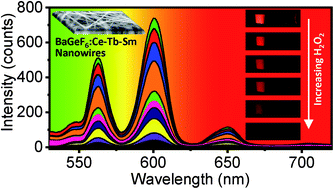
Fluorides are promising host materials for optical applications. This paper reports the photoluminescent (PL) and cathodoluminescent (CL) characteristics of barium hexafluorogermanate BaGeF6 nanowires codoped with Ce3+, Tb3+ and Sm3+ rare earth ions, produced by a solvothermal route. The synthesized BaGeF6 nanowires exhibit uniform morphology and size distribution. X-ray diffraction divulges the one-dimensional growth of crystalline BaGeF6 structure, with the absence of any impurity phases. Visible luminescence is recorded from the nanowires in green and red regions, when the nanowires are codoped with Ce3+/Tb3+, and Ce3+/Tb3+/Sm3+, respectively, under a UV excitation source. The PL emission from the codoped BaGeF6 nanowires, when excited by a 254 nm source, originates from the efficient energy transfer bridges between Ce3+, Tb3+ and Sm3+ ions. The decay time of the visible luminescent emission from the nanowires is in the order of subnanoseconds, being one of the shortest decay time records from inorganic scintillators. The CL emission from the BaGeF6 nanowires in the tunable visible range reveals their potential use for the detection of high-energy radiation. The PL emissions are sensitive to H2O2 at low concentrations, enabling their high-sensitivity detection of H2O2 using BaGeF6 nanowires. A comparison with BaSiF6 nanowires is made in terms of decay time and its sensitivity towards H2O2.


 Please wait while we load your content...
Please wait while we load your content...