Low temperature synthesis of BiFeO3 nanoparticles with enhanced magnetization and promising photocatalytic performance in dye degradation and hydrogen evolution†
Abstract
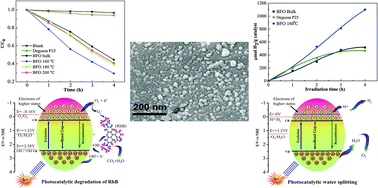
In this investigation, we have synthesized BiFeO3 nanoparticles by varying hydrothermal reaction temperatures from 200 °C to 120 °C to assess their visible-light driven photocatalytic activity along with their applicability for hydrogen production via water splitting. The rhombohedral perovskite structure of BiFeO3 is formed for hydrothermal reaction temperature up to 160 °C. However, for a further decrement of hydrothermal reaction temperature a mixed sillenite phase is observed. The XRD Rietveld analysis, XPS analysis and FESEM imaging ensure the formation of single-phase and well crystalline nanoparticles at 160 °C reaction temperature with 20 nm of average size. The nanoparticles fabricated at this particular reaction temperature also exhibit improved magnetization, reduced leakage current density and excellent ferroelectric behavior. These nanoparticles demonstrate considerably high absorbance in the visible range with a low band gap (2.1 eV). The experimentally observed band gap is in excellent agreement with the calculated band gap using first-principles calculations. The favorable photocatalytic performance of these nanoparticles has been able to generate more than two times of solar hydrogen compared to that produced by bulk BiFeO3 as well as commercially available Degussa P25 titania. Notably, the experimentally observed band gap is almost equal for both bulk material and nanoparticles prepared at different reaction temperatures. Therefore, in solar energy applications, the superiority of BiFeO3 nanoparticles prepared at 160 °C reaction temperature may be attributed not only to their band gap but also to other factors, such as reduced particle size, excellent morphology, good crystallinity, large surface to volume ratio, ferroelectricity and so on.


 Please wait while we load your content...
Please wait while we load your content...