Antimicrobial synergy of cationic grafted poly(para-phenylene ethynylene) and poly(para-phenylene vinylene) compounds with UV or metal ions against Enterococcus faecium
Abstract
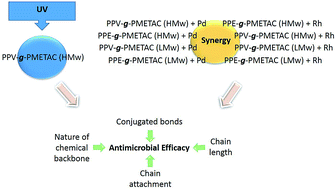
The rise in multidrug resistant bacteria is an area of growing concern and it is essential to identify new biocidal agents. Cationic grafted compounds were investigated for their antimicrobial properties using minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) tests. Synergy testing was carried out using the compounds in the presence of ultraviolet (UV). Fractional inhibitory concentration (FIC) and fractional bactericidal concentration (FBC) tests were carried out using the cationic molecules in conjunction with metal ion solutions of gold, silver, palladium, platinum, rhodium, titanium, tin, vanadium and molybdenum. Individually, the cationic compounds containing quaternary amines, polyphenylene vinylene (PPV) with long polyacrylate grafts (PPV-g-PMETAC (HMw)), polyphenylene ethylene (PPE) with long polyacrylate grafts (PPE-g-PMETAC (HMw)), polyphenylene vinylene (PPV) with short polyacrylate grafts (PPV-g-PMETAC (LMw)) and polyphenylene ethylene (PPE) with short polyacrylate grafts (PPE-g-PMETAC (LMw)) were effective against Enterococcus faecium. The most successful compound under UV was PPV-g-PMETAC (HMw). Following the FICs, palladium and rhodium ion solutions caused a synergistic reaction with all four tested compounds. The presence of conjugated bonds in the cationic molecules increased its antimicrobial activity. These results suggest that the chemical backbone of the compounds, alongside the chain lengths and chain attachment affect the antimicrobial efficacy of a compound. These factors should be taken into consideration when formulating new biocidal combinations.


 Please wait while we load your content...
Please wait while we load your content...