Discovery and preliminary SAR of 14-aryloxy-andrographolide derivatives as antibacterial agents with immunosuppressant activity†
Abstract
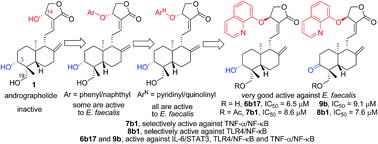
Antibacterials (which restore gut flora balance) and immunosuppressants (which correct immune defects) are two important and effective therapeutic agents for the treatment of inflammatory bowel disease (IBD) in clinical use today. Since the structural skeleton of andrographolide, isolated from Andrographis paniculata, has become known as a natural antibiotic with anti-inflammation and heat-clearing and detoxifying properties, 14-aryloxy andrographolide derivatives have been designed, synthesized, and tested for their antibacterial effects on E. coli, S. aureus, and E. faecalis, which are related to IBD. It has been discovered in this study that the andrographolide skeleton is more selective against E. faecalis, the 14-aryloxy group with basicity is important for antibacterial functions, and the 14-(8′-quinolinyloxy) group is a good pharmacophore with antibacterial activity. In addition, we found that 7b1 and 8b1 are good and selective inhibitors of E. faecalis; two 14β-(8′-quinolinyloxy) andrographolide derivatives, 6b17 and 9b, exhibit good activity against E. coli, S. aureus, and E. faecalis. Likewise and importantly, further exploration of immunosuppressant activity for IBD shows that compound 7b1 is a selective inhibitor of the TNF-α/NF-κB signaling pathway, whereas 8b1 is selectively active against the TLR4/NF-κB signaling pathway; moreover, the compounds 6b17 and 9b are active in inhibiting the IL-6/STAT3, TLR4/NF-κB, and TNF-α/NF-κB signaling pathways. Based on these results, we have further focused on the development of dual function inhibitors of IBD as antibacterial and immunosuppressant agents by structural modification of andrographolide.
- This article is part of the themed collection: Editors' collection: Chemical Biology


 Please wait while we load your content...
Please wait while we load your content...