Pyridazine-bridged cationic diiridium complexes as potential dual-mode bioimaging probes†
Abstract
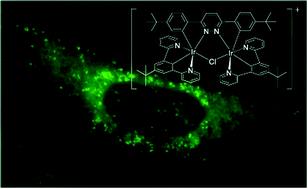
A novel diiridium complex [(N^C^N)2Ir(bis-N^C)Ir(N^C^N)2Cl]PF6 (N^C^N = 2-[3-tert-butyl-5-(pyridin-2-yl)phenyl]pyridine; bis-N^C = 3,6-bis(4-tert-butylphenyl)pyridazine) was designed, synthesised and characterised. The key feature of the complex is the bridging pyridazine ligand which brings two cyclometallated Ir(III) metal centres close together so that Cl also acts as a bridging ligand leading to a cationic complex. The ionic nature of the complex offers a possibility of improving solubility in water. The complex displays broad emission in the red region (λem = 520–720 nm, τ = 1.89 μs, Φem = 62% in degassed acetonitrile). Cellular assays by multiphoton (λex = 800 nm) and confocal (λex = 405 nm) microscopy demonstrate that the complex enters cells and localises to the mitochondria, demonstrating cell permeability. Further, an appreciable yield of singlet oxygen generation (ΦΔ = 0.45, direct method, by 1O2 NIR emission in air equilibrated acetonitrile) suggests a possible future use in photodynamic therapy. However, the complex has relatively high dark toxicity (LD50 = 4.46 μM), which will likely hinder its clinical application. Despite this toxicity, the broad emission spectrum of the complex and high emission yield observed suggest a possible future use of this class of compound in emission bioimaging. The presence of two heavy atoms also increases the scattering of electrons, supporting potential future applications as a dual fluorescence and electron microscopy probe.
- This article is part of the themed collection: Editors’ collection: Photodynamic therapy


 Please wait while we load your content...
Please wait while we load your content...