Hyaluronan-based graft copolymers bearing aggregation-induced emission fluorogens†
Abstract
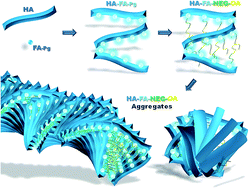
In order to develop a technology platform based on two natural compounds from biorenewable resources, a short series of hyaluronan (HA) copolymers grafted with propargylated ferulic acid (HA–FA–Pg) were designed and synthesized to show different grafting degree values and their optical properties were characterized in comparison with reference compounds containing the same ferulate fluorophore. Interestingly, these studies revealed that the ferulate fluorophore was quite sensitive to the restriction of intramolecular motion and its introduction into the rigid HA backbone, as in HA–FA–Pg graft copolymers, led to higher photoluminescence quantum yield values than those obtained with the isolated fluorophore. Thus, the propargyl groups of HA–FA–Pg derivatives were exploited in the coupling with oleic acid through a biocompatible nona(ethylene glycol) spacer as an example of the possible applications of this technology platform. The resulting HA–FA–NEG–OA materials showed self-assembling capabilities in aqueous environment. Furthermore, HA–FA–NEG–OA derivatives have been shown to interact with phospholipid bilayers both in liposomes and living cells, retaining their fluorogenic properties and showing a high degree of cytocompatibility and for this reason they were proposed as potential biocompatible self-assembled aggregates forming new materials for biomedical applications.


 Please wait while we load your content...
Please wait while we load your content...