Reversible ionically-crosslinked single chain nanoparticles as bioinspired and recyclable nanoreactors for N-heterocyclic carbene organocatalysis†
Abstract
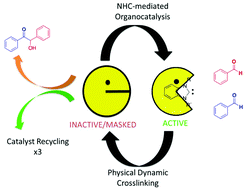
The intrinsic advantages of poly(ionic liquid)s (PILs), based on their high chemical activity and flexible structure, have been harnessed by exploring their applicability as catalytic single chain nanoparticles (SCNPs). A non-covalent bioinspired approach has been established to ionically crosslink an imidazolium-based poly(ionic liquid) to form folded SCNPs. An amphiphilic styrenic-type coPIL was synthesized by reversible addition fragmentation chain transfer (RAFT) to include hydrophilic stabilizer units, hydrophobic spacers and two antagonist functionalities randomly distributed through the polymer backbone. The antagonist functionalities were then intramolecularly and ionically crosslinked using a simple anion metathesis reaction, which resulted in folding to form the SCNPs under mild conditions. The folding process enabled the protection of the N-heterocyclic carbene (NHC) functionality, through the benzoate–imidazolium interaction between antagonist monomer units. Upon the application of heat, free NHCs could be generated within the confined SCNPs, which could be further utilized in benzoin catalysis. Most importantly, the reversible nature of the crosslinking and reversible generation of the active functionality allowed for the utilization of the SCNPs as a recyclable catalytic support.


 Please wait while we load your content...
Please wait while we load your content...