Grafting-from lipase: utilization of a common amino acid residue as a new grafting site†
Abstract
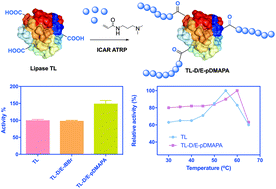
Protein–polymer hybrids are used in a variety of fields including catalysis, detection, and therapeutics. The grafting-from method for the synthesis of these biohybrids has gained popularity due to the ease of synthesis and purification. In this method, an initiator or chain transfer agent (CTA) is ligated onto an amino acid residue, typically lysine or cysteine, and polymers are subsequently grown in situ. In this manuscript, we report the preparation of protein polymer hybrids by grafting-from a previously overlooked acidic amino acid residue (glutamic and aspartic acid) and compare our results to protein polymer hybrids, grafted from the traditional lysine residue. Herein, we conjugated an atom transfer radical polymerization (ATRP) initiator to acidic amino acid residues and lysine residues and grew polymers from Thermomyces lanuginosa lipase (TL). N-[3-(N,N-Dimethylamino)propyl] acrylamide was grafted from the TL initiator, and the enzymatic activity of protein polymer hybrids was compared. We found that the acidic residues are easily modified with multiple ATRP initiators and polymers are readily grown. Additionally, the hybrids grafted from acidic residues demonstrated a 50% increase in enzyme activity compared to those grafted from lysine residues. Moreover, the activity was higher than that of native lipase TL in both cases. The polymers that were grafted-from the acid residues tended to provide the hybrids with a higher activity at elevated temperatures. These results point to a new amino acid ligation strategy for preparing protein polymer hybrids via a grafting-from method.


 Please wait while we load your content...
Please wait while we load your content...