Polypeptide gels incorporating the exotic functional aromatic amino acid 4-amino-l-phenylalanine†
Abstract
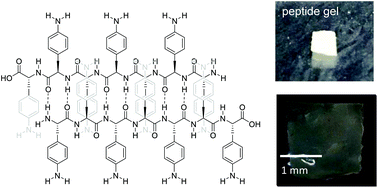
High-molecular-weight polypeptides with functional aromatic side chains, poly(4-amino-L-phenylalanine), 4APhe, were prepared by the metal-initiated polymerization of the Nα-carboxyanhydride of the corresponding amino acid, which is a microbial derivative of phenylalanine. The polypeptide P4APhe has good water solubility as opposed to those of the conventional aromatic poly(amino acid)s such as poly(L-phenylalanine) or poly(L-tyrosine). On the other hand, P4APhe shows an in situ gelation ability upon the swelling of dried samples in solvents, due to the formation of crystalline β-sheet domains that work as cross-linking junctions. Moreover, the cationic copolymers of 4APhe with L-lysine showed a pH-responsive gelling behavior resulting from changes in the amphipathic balance of crystalline β-strands, α-helical structures, and hydrophilic random coils. Notably, small amounts of 4APhe units in copolypeptides with L-lysine were found to enhance adoption α-helical conformations.


 Please wait while we load your content...
Please wait while we load your content...