Accelerating the acidic degradation of a novel thermoresponsive polymer by host–guest interaction†
Abstract
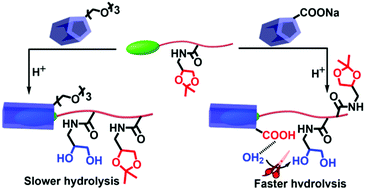
We synthesized a novel thermoresponsive and pH-degradable polymer, (poly(N-[(2,2-dimethyl-1,3-dioxolane)methyl]acrylamide)), with a pyridinium salt based end group, which can form a host–guest complex with various pillar[5] arenes. This complex formation led to an increase of the LCST of the polymer whereat carboxylate functionalized pillar[5] arenes caused a stronger shift than the respective oligoethylene oxide modified ones. This trend is found to be coherent with the strength of interaction of the respective host and guest, which was analyzed by NMR studies and isothermal titration calorimetry (ITC). Further investigations on this complex revealed that the hydrolysis of the polymer can be accelerated in the presence of the carboxylate based pillar arene under acidic conditions, which was not the case for the oligoethylene oxide modified system or a structurally similar carboxylic acid unit not forming a complex. The host–guest interaction therefore not only represents a versatile approach to tune the solubility behavior of polymers, but also influences pH dependent degradability.


 Please wait while we load your content...
Please wait while we load your content...