Self-stabilized, hydrophobic or PEGylated paclitaxel polymer prodrug nanoparticles for cancer therapy†
Abstract
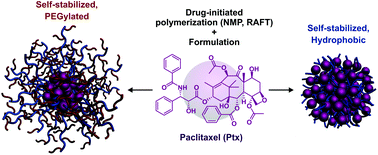
Facile derivatization of paclitaxel (Ptx) and subsequent “drug-initiated” synthesis of well-defined Ptx-polymer prodrugs was performed from nitroxide-mediated polymerization or reversible addition–fragmentation chain transfer polymerization. Short polyisoprene (Mn = 2640–4310 g mol−1, Đ ∼ 1.1) or poly[(oligo(ethylene glycol) methyl ether methacrylate)] (Mn = 5580–7530 g mol−1, Đ ∼ 1.2) chains were grown from Ptx in a controlled fashion and enabled, for the first time, the formation of either self-stabilized, all-hydrophobic Ptx-polymer prodrug nanoparticles or their PEGylated counterparts. They exhibited average diameters in the 110–235 nm range, great colloidal stability in PBS and cell culture medium, high drug loadings (up to 32 wt%) and cytotoxicity similar to that of the parent drug on three different cancer cell lines (A549, L1210 and MCF-7). This versatile approach is expected to provide more potent anticancer nanocarriers and rejuvenate the development of Ptx-based nanomedicines.


 Please wait while we load your content...
Please wait while we load your content...