A simple, modular synthesis of bifunctional peptide-polynorbornenes for apoptosis induction and fluorescence imaging of cancer cells†
Abstract
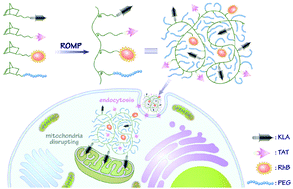
Bifunctional peptide-polynorbornenes, comprised of four modules, D-enantiomeric, protease-resistant sequence D[KLAKLAK]2 peptide as the anticancer motif, cell-penetrating peptide TAT sequence as the “transmission adapter”, red fluorescence RhB as the imaging reporter, and biocompatible PEG as the hydrophilic solubilizer, were designed and fabricated via a simple ROMP (ring-opening metathesis polymerization) strategy. Attributed to the integration of both KLA and TAT peptides, these bioactive copolymers exhibit the synergistic induction of tumor cell apoptosis. Confocal laser scanning microscopy and flow cytometric analyses reveal that these bifunctional peptide KLA–TAT copolymers target or act on mitochondria and disrupt the mitochondrial membrane potential (MMP) after efficient internalization with the aid of the cell-penetrating TAT motif, thus inducing mitochondrial-dependent apoptosis of cancer cells with enhanced activity. More importantly, this modularly designed ROMP strategy could integrate the multi-functionalities of targeting, imaging and cancer therapeutics into a single polymeric scaffold, and endow synthetic polymers with tremendous potential to serve as a new type of agent for cancer diagnostics and treatment.


 Please wait while we load your content...
Please wait while we load your content...