Photochromic and luminescence properties of a hybrid compound based on indoline spiropyran of the coumarin type and azomethinocoumarin
Abstract
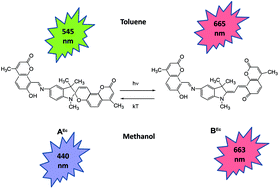
A novel hybrid compound whose molecule includes the photochromic spiropyran fragment and two fluorophores – coumarin and salicylideneimine – was synthesized by the reaction of two equivalents of 8-formyl-7-hydroxy-4-methylcoumarin with 5-amino-1,3,3-trimethyl-2-methyleneindoline. Its spectral-kinetic and fluorescence properties were studied in comparison with model compounds – 7-hydroxy-4-methyl-coumarin, spiropyran of the coumarin type, and azomethinocoumarin in toluene and ethanol. It was shown that the hybrid compound exhibited photochromic transformations in both solvents. Different fluorescence emissions of the initial spirocycle and UV irradiation-induced merocyanine forms were observed. One green fluorescence emission with a maximum at 545 nm was observed for the initial closed form in toluene, and several emissions with maxima at 390 nm, 440 nm, 450 nm (violet), and 530 nm (green) appeared in ethanol. An additional red emission of the open merocyanine form in the region of 665 nm appeared after UV irradiation in both solvents. Reversible modulation of fluorescence in both the initial and photoinduced merocyanine forms in the process of photochromic transformations was discovered. It was shown that the fluorescence modulation of the hybrid compound in weakly polar toluene was caused by resonance energy transfer from the fluorescent fragment of the photochromic compound to the photoinduced merocyanine form, leading to the process of photochromic transformations. Reversible modulation of fluorescence in ethanol solutions was achieved through a photoinduced change in the ratio of the concentrations of conformers.


 Please wait while we load your content...
Please wait while we load your content...