Asymmetric transfer hydrogenation of cycloalkyl vinyl ketones to allylic alcohols catalyzed by ruthenium amido complexes†
Abstract
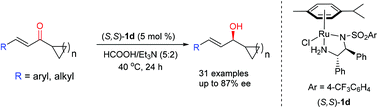
A chemoselective 1,2-reduction of cycloalkyl vinyl ketones via asymmetric transfer hydrogenation is described. The reduction proceeded smoothly with a chiral diamine ruthenium complex as a catalyst and a HCOOH–NEt3 azeotrope as both a hydrogen source and solvent under mild conditions. A wide range of 1-cycloalkyl chiral allylic alcohols were obtained in good yields and up to 87% ee. It was found that the alkyl group plays an important role in the enantioselectivity.
- This article is part of the themed collections: Synthetic methodology in OBC and Catalysis & biocatalysis in OBC


 Please wait while we load your content...
Please wait while we load your content...