Decarbonylative cross-coupling of amides
Abstract
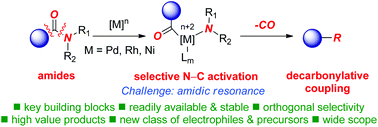
Cross-coupling reactions are among the most powerful C–C and C–X bond forming tools in organic chemistry. Traditionally, cross-coupling methods rely on the use of aryl halides or pseudohalides as electrophiles. In the past three years, decarbonylative cross-couplings of amides have emerged as an attractive method for the construction of a wide variety of carbon–carbon and carbon–heteroatom bonds, allowing for the synthetically-valuable functional group inter-conversion of the amide bond. These previously elusive reactions hinge upon selective activation of the N–C(O) acyl amide bond, followed by CO extrusion, in a formal double N–C/C–C bond activation, to generate a versatile aryl–metal intermediate as an attractive alternative to traditional cross-couplings of aryl halides and pseudohalides. In this perspective review, we present recent advances and key developments in the field of decarbonylative cross-coupling reactions of amides as well as discuss future challenges and potential applications for this exciting field.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...