Tetraphenylethylene conjugated p-hydroxyphenacyl: fluorescent organic nanoparticles for the release of hydrogen sulfide under visible light with real-time cellular imaging†
Abstract
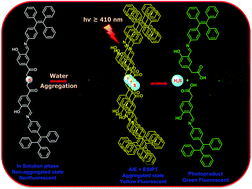
Hydrogen sulfide (H2S) behaves like a two-edged sword, at low concentrations it has beneficial and cytoprotective effects, while at higher concentrations it exhibits toxicity. Hence there is a keen interest in developing light responsive H2S donors with a spatio-temporal controlled release. Herein, we report visible light activatable tetraphenylethylene conjugated p-hydroxyphenacyl (TPE-pHP-H2S) nanoparticles for the release of hydrogen sulfide (H2S) with a real time monitoring ability. Our newly designed photoresponsive single component organic nanoparticle based H2S donor is built by integrating the tetraphenylethylene (TPE) moiety and p-hydroxyphenacyl (pHP) group so that it can display both aggregation-induced emission (AIE) and excited state intramolecular proton transfer (ESIPT) properties. Aggregation-induced emission enhancement was exhibited by our TPE-pHP-H2S NP donor, which was then explored for the cellular imaging application. The ESIPT by the pHP moiety provided unique advantages to our TPE-pHP-H2S NP donor which include (i) the excitation wavelength extended to >410 nm (ii) a large Stokes shift (iii) a low inner filter effect and (iv) real-time monitoring of H2S release by a simple fluorescent colour change. In vitro studies showed that the TPE-pHP-H2S NP donor presents excellent properties like real-time monitoring, photoregulated H2S release and biocompatibility.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...