Asymmetric total synthesis of naturally occurring spirocyclic tetranorsesquiterpenoid lanceolactone A†
Abstract
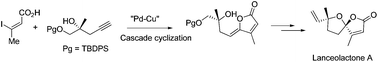
Asymmetric total synthesis of naturally occurring γ-butenolide containing [4.4]spiro-tetrahydrofuran lanceolactone A has been reported in the present work. Bimetallic (“Pd–Cu”) cascade cyclization was the crucial reaction employed for the construction of the γ-butenolide framework of the natural product. Subsequently, iodocyclization and reductive deiodination through a transfer hydrogenation reaction were applied to access the target molecule in an efficient manner.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...