Toward aplyronine payloads for antibody–drug conjugates: total synthesis of aplyronines A and D†
Abstract
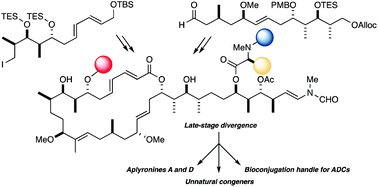
The aplyronines are a family of antimitotic marine macrolides that disrupt cytoskeletal dynamics by dual targeting of both actin and tubulin. Given their picomolar cytotoxicity profile and unprecedented mode of action, the aplyronines represent an excellent candidate as a novel payload for the development of next-generation antibody–drug conjugates (ADCs) for cancer chemotherapy. Enabled by an improved second-generation synthesis of the macrolactone core 5, we have achieved the first total synthesis of the most potent congener aplyronine D together with a highly stereocontrolled synthesis of aplyronine A. To facilitate step economy, an adventurous site-selective esterification of the C7 hydroxyl group was performed to install the N,N,O-trimethylserine pharmacophore to directly afford aplyronines A and D. Toward the assembly of ADCs incorporating an aplyronine warhead, the C29-ester derivative 4 featuring an Fmoc-amino substituted linker attached to the actin-binding tail region was also prepared by adapting this flexible endgame.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...