Light-responsive bicyclic peptides†
Abstract
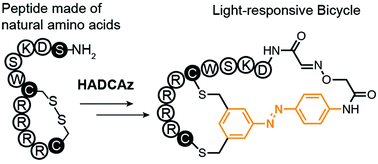
In this paper, we describe a method for the synthesis of light-responsive (LR) bicyclic macrocycles from linear peptides composed of 20 natural amino acids. Small molecules, peptide macrocycles, and protein conjugates that reversibly turn their function on and off in response to visible light enabled the fields of photopharmacology and optochemical genetics. Bioactive LR molecules could be produced by grafting azobenzene or other LR-structures onto molecules with known biological functions (e.g., alpha-helical peptides). It is also possible to discover such LR ligands de novo by selecting compounds with a desired function—such as binding to a target—from a library of LR-compounds or a genetically-encoded (GE) library of LR-macrocycles. The bicyclic topology of ligands offers added value such as improved binding and stability when compared to monocyclic peptides, but approaches for the design of bicyclic light-responsive architectures are limited. To address this need, we developed a tridentate C2-symmetric hydroxyl amine and di-chlorobenzene containing azobenzene (HADCAz) LR-linker with two orthogonally reactive functionalities (chlorobenzyl and hydroxylamine) to convert a linear unprotected peptide into a bicyclic peptide in a one-pot, two-step reaction. This linker reversibly isomerizes from the trans to cis form upon irradiation with blue light (365 nm). The resulting bicyclic peptide contains two loops of amino acids, one of which is constrained with an azobenzene moiety that can change the conformation in response to visible light. A scalable synthetic route to the HADCAz linker allowed us to demonstrate its application in multiple synthetic bicyclic peptides with loops that contain 2–5 amino acids.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...