Synergy of light harvesting and energy transfer as well as short-range charge shift reactions in multicomponent conjugates†
Abstract
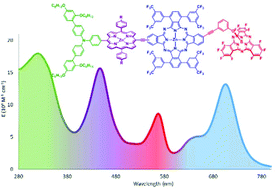
We report herein on the design, the synthesis, and the characterization of a panchromatic, charge stabilizing electron donor–acceptor conjugate: (BBPA)3–ZnPor–ZnPc–SubPc 1. Each component, that is, bis(biphenyl)phenylamine (BBPA), Zn(II) porphyrin (ZnPor), Zn(II) phthalocyanine ZnPc, and subphthalocyanine (SubPc), has been carefully chosen and modified to enable a cascade of energy and charge transfer processes. On one hand, ZnPor, has been functionalized with three electron-donating BBPA as primary and secondary electron donors and to stabilize the final charge-separated state, and, on the other hand, a perfluorinated SubPc has been selected as ultimate electron acceptor. In addition, the ZnPc unit contains several trifluoromethylphenyl moieties to match its energy levels to those of the other components. In fact, irradiation of the heteroarray 1 triggers a cascade of light harvesting across the entire visible range, unidirectional energy transfer, exergonic charge separating, and short-range charge shifting to afford in 14% quantum yield a (BBPA)3˙+–ZnPor–ZnPc–SubPc˙− charge-separated state. The lifetime of the latter reaches well into the range of tens of nanoseconds.


 Please wait while we load your content...
Please wait while we load your content...