Occupational exposure to graphene based nanomaterials: risk assessment
Abstract
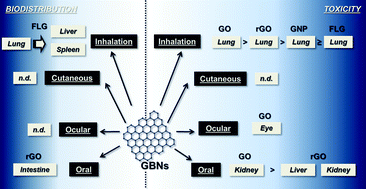
Graphene-based materials (GBMs) are a family of novel materials including graphene, few layer graphene (FLG), graphene oxide (GO), reduced graphene oxide (rGO) and graphene nanoplatelets (GNP). Currently, the risk posed by them to human health is associated mainly with the occupational exposure during their industrial and small-scale production or waste discharge. The most significant occupational exposure routes are inhalation, oral, cutaneous and ocular, inhalation being the majorly involved and most studied one. This manuscript presents a critical up-to-date review of the available in vivo toxicity data of the most significant GBMs, after using these exposure routes. The few in vivo inhalation toxicity studies (limited to 5-days of repeated exposure and only one to 5 days per week for 4 weeks) indicate inflammatory/fibrotic effects at the pulmonary level, not always reversible after 14/90 days. More limited in vivo data are available for the oral and ocular exposure routes, whereas the studies on cutaneous toxicity are at the initial stage. A long persistence of GBMs in rodents is recorded, while contradictory genotoxic data are reported. Data gap identification is also provided. Based on the available data, the occupational exposure limit cannot be determined. More experimental toxicity studies according to specific guidelines (tentatively validated for nanomaterials) and more information on the actual occupational exposure level to GBMs are needed. Furthermore, ADME (Absorption, Distribution, Metabolism, Excretion), genotoxicity, developmental and reproductive toxicity data related to the occupational exposure to GBMs have to be implemented. In addition, sub-chronic and/or chronic studies are still needed to completely exclude other toxic effects and/or carcinogenicity.
- This article is part of the themed collections: Recent Review Articles, Graphene Turns 15: Bio-implications and Bio-applications and Nanoscale 10th Anniversary: Top Authors


 Please wait while we load your content...
Please wait while we load your content...