Highly stable photoelectrochemical cells for hydrogen production using a SnO2–TiO2/quantum dot heterostructured photoanode†
Abstract
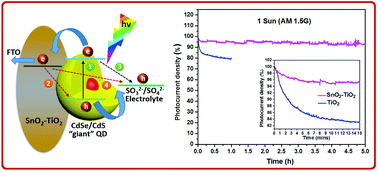
Photoelectrochemical (PEC) water splitting implementing colloidal quantum dots (QDs) as sensitizers is a promising approach for hydrogen (H2) generation, due to the QD's size-tunable optical properties. However, the challenge of long-term stability of the QDs is still unresolved. Here, we introduce a highly stable QD-based PEC device for H2 generation using a photoanode based on a SnO2–TiO2 heterostructure, sensitized by CdSe/CdS core/thick-shell “giant” QDs. This hybrid photoanode architecture leads to an appreciable saturated photocurrent density of ∼4.7 mA cm−2, retaining an unprecedented ∼96% of its initial current density after two hours, and sustaining ∼93% after five hours of continuous irradiation under an AM 1.5G (100 mW cm−2) simulated solar spectrum. Transient photoluminescence (PL) measurements demonstrate that the heterostructured SnO2–TiO2 photoanode exhibits faster electron transfer compared with the bare TiO2 photoanode. The lower electron transfer rate in the TiO2 photoanode can be attributed to slow electron kinetics in the ultraviolet regime, revealed by ultrafast transient absorption spectroscopy. Graphene microplatelets were further introduced into the heterostructured photoanode, which boosted the photocurrent density to ∼5.6 mA cm−2. Our results demonstrate that the SnO2–TiO2 heterostructured photoanode holds significant potential for developing highly stable PEC cells.


 Please wait while we load your content...
Please wait while we load your content...