Revealing the principal attributes of protein adsorption on block copolymer surfaces with direct experimental evidence at the single protein level†
Abstract
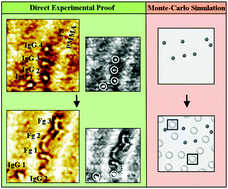
Understanding protein adsorption onto polymer surfaces is of great importance in designing biomaterials, improving bioanalytical devices, and controlling biofouling, to name a few examples. Although steady research efforts have been advancing this field, our knowledge of this ubiquitous and complex phenomenon is still limited. In this study, we elucidate competitive protein adsorption behaviors sequentially occurring onto nanoscale block copolymer (BCP) surfaces via combined experimental and computer simulation approaches. The model systems chosen for our investigation are immunoglobulin G and fibrinogen introduced in different orders into the self-assembled nanodomains of poly(styrene)-block-poly(methylmethacrylate). We unambiguously reveal the adsorption, desorption, and replacement events of the same protein molecules via single protein tracking with atomic force microscopy. We then ascertain adsorption-related behaviors such as lateral mobility and self-association of proteins. We provide the much-needed, direct experimental proof of sequential adsorption events at the biomolecular level, which was virtually nonexistent before. We determine key protein adsorption pathways and dominant tendencies of sequential protein adsorption. We also reveal preadsorbed surface-associated behaviors in sequential adsorption, distinct from situations involving initially empty surfaces. We perform Monte-Carlo simulations to further substantiate our experimental outcomes. Our endeavors in this study may facilitate a well-guided mechanistic understanding of protein–polymer interactions by providing definite experimental evidence of competitive, sequential adsorption at the nanoscale. Increasingly, biomaterial and biomedical applications rely on systems of multicomponent proteins and chemically intricate, nanoscale polymer surfaces. Hence, our findings can also be beneficial for the development of next-generation nanobiomaterials and nanobiosensors exploiting self-assembled BCP nanodomain surfaces.


 Please wait while we load your content...
Please wait while we load your content...