Marrying SPR excitation and metal–support interactions: unravelling the contribution of active surface species in plasmonic catalysis†
Abstract
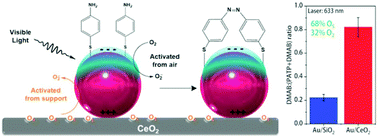
Plasmonic catalysis takes advantage of the surface plasmon resonance (SPR) excitation to drive or accelerate chemical transformations. In addition to the plasmonic component, the control over metal–support interactions in these catalysts is expected to strongly influence the performances. For example, CeO2 has been widely employed towards oxidation reactions due to its oxygen mobility and storage properties, which allow for the formation of Ce3+ sites and adsorbed oxygen species from metal–support interactions. It is anticipated that these species may be activated by the SPR excitation and contribute to the catalytic activity of the material. Thus, a clear understanding of the role played by the SPR-mediated activation of surface oxide species at the metal–support interface is needed in order to take advantage of this phenomenon. Herein, we describe and quantify the contribution from active surface oxide species at the metal–support interface (relative to O2 from air) to the activities in green SPR-mediated oxidation reactions. We employed CeO2 decorated with Au NPs (Au/CeO2) as a model plasmonic catalyst and the oxidation of p-aminothiophenol (PATP) and aniline as proof-of-concept transformations. We compared the results with SiO2 decorated with Au NPs (Au/SiO2), in which the formation of surface oxide species at the metal–support interface is not expected. We found that the SPR-mediated activation of surface oxide species at the metal–support interface in Au/CeO2 played a pivotal role in the detected activities, being even higher than the contribution coming from the activation of O2 from air.
- This article is part of the themed collection: Materials and Nano Research in Brazil


 Please wait while we load your content...
Please wait while we load your content...