Protein–protein interactions in polyketide synthase–nonribosomal peptide synthetase hybrid assembly lines
Abstract
Covering: up to early 2018
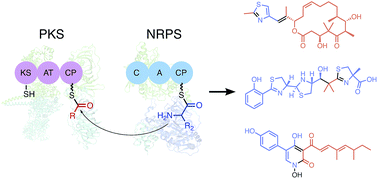
Polyketides and nonribosomal peptides are two major families of natural product with a broad range of biological activities. Polyketide synthases (PKSs) assemble small acetic acid-type acyl building blocks into polyketides through C–C bonds, and nonribosomal peptide synthetases (NRPSs) assemble amino acids into peptides through amide bonds. PKS–NRPS hybrid assembly lines build structurally complex polyketide–amino acid/peptide hybrid molecules that incorporate both acyl and aminoacyl building blocks into their products. Their combined functionalities expand the biological activities of these molecules by mixing their chemical properties. Protein–protein interactions are necessary within PKS–NRPS hybrid assembly lines to achieve accurate linkage between the PKS and NRPS systems. This review summarizes the current understanding of the roles and importance of the protein–protein interactions in various PKS–NRPS hybrid assembly lines.
- This article is part of the themed collection: Understanding biosynthetic protein-protein interactions


 Please wait while we load your content...
Please wait while we load your content...