Synthesis of a SN38 prodrug grafted to amphiphilic phosphorylcholine polymers and their prodrug miceller properties†
Abstract
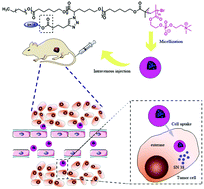
Polymer prodrug micelles, combining the advantages of prodrugs and polymer micelles, can greatly improve the solubility, permeability and stability of drugs. In this work, amphiphilic block copolymers poly(α-azide caprolactone-co-caprolactone)-b-poly(2-methacryloyloxyethyl phosphorylcholine), P(ACL-co-CL)-b-PMPC, were synthesized, and an antineoplastic drug 7-ethyl-10-hydroxy camptothecin (SN38) was grafted to the amphiphilic copolymers to obtain a prodrug and their micelles. The preparation of P(ACL-co-CL)-b-PMPC was proceeded by ring-opening polymerization of ε-caprolactone/α-bromide caprolactone, converting the bromide to an azide, and atom transfer radical polymerization (ATRP). The camptothecin derivative SN38-alkyne was bonded to the polymer to obtain P(CL/CL-g-SN38)-b-PMPC by a “click reaction”. The obtained polymer–drug complex can easily self-assemble to form prodrug micelles with the shell composed of PMPC and the core composed of PCL and SN38. The micelles presented characteristics including improved drug loading efficiency and stabilization of the drug. In vivo, the retention time of the prodrug micelles in blood could be tripled, and the clearance was delayed compared with the SN38 solution. The in vivo anti-tumor experiments demonstrated that the prodrug micelles had a better treatment effect and low toxicity to 4T1-bearing BALB/C mice.


 Please wait while we load your content...
Please wait while we load your content...