Trimetallic PtPdCu nanowires as an electrocatalyst for methanol and formic acid oxidation
Abstract
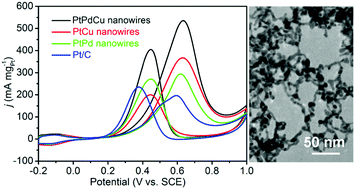
In this work, trimetallic PtPdCu nanowires with a polycrystalline structure are prepared by a one-pot solvothermal method. It is found that the presence of polyvinylpyrrolidone and KBr is essential for the formation of nanowire morphology. The electrocatalytic performances of the PtPdCu nanowires toward methanol oxidation reaction and formic acid oxidation reaction in an acidic medium are tested. The trimetallic PtPdCu nanowires show better electrocatalytic activity and stability than bimetallic PtCu and PtPd nanowires and commercial Pt/C catalyst. The better electrocatalytic activities of the PtPdCu nanowires can be explained by the electronic, composition and morphological effects. The Pd and Cu atoms in the PtPdCu nanowires modulate the electronic structure of Pt atoms, thus weakening the CO adsorption energy on Pt. The Pd atoms also promote CO oxidation on the Pt surface. The ultrathin polycrystalline nanowire morphology affords a large quantity of active sites for electrocatalysis reactions. The PtPdCu nanowires may have potential application as a promising anode catalyst for fuel cells.


 Please wait while we load your content...
Please wait while we load your content...