A fullerene helical peptide: synthesis, characterization and formation of self-assembled monolayers on gold surfaces†
Abstract
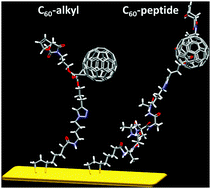
The synthesis of a C60-peptide using “clickable” fullerene and peptide derivatives is described. The peptide is composed of a repeating sequence of α-aminoisobutyric acid (Aib) and two L-alanine (Ala) residues, promoting the formation of a helical conformation, which has been confirmed by IR absorption, NMR and circular dichroism measurements. In addition, the presence of a lipoyl moiety, at the end of the peptide sequence, allows the formation of self-assembled monolayers of the C60-peptide and the parent peptide on a gold surface. A C60-alkyl derivative was also prepared to compare the self-assembly properties of fullerene derivatives containing peptides or alkyl chains. The fullerene assembly on a gold substrate characterized by quartz crystal microbalance and by cyclic voltammetry show that the monolayers containing alkyl chains are slightly less well packed than the peptide monolayers. Finally, polarization modulation reflection adsorption infra-red spectroscopy measurements indicate that the C60-peptide tends to be more vertical than the parent peptide which could originate from complementary C60–C60 and helical–helical interactions.


 Please wait while we load your content...
Please wait while we load your content...