Adjusting the energy levels and bandgaps of conjugated polymers via Lewis acid–base reactions†
Abstract
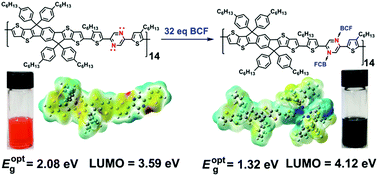
Lewis acid–base chemistry has been demonstrated to be an effective strategy to adjust the optoelectronic properties of organic materials, especially conjugated small molecules. However, for conjugated polymers with large molecular weight, the stoichiometry of the Lewis acid–base reaction remains an interesting topic to research. In this work, two copolymers, namely P1 and P2, were synthesized, by covalently bonding two fused and co-planar units, i.e., indacenodithiophene (IDT) and indacenodithieno[3,2-b]thiophene (IDTT), respectively, with a pyrazine unit, a strong Lewis base containing two nitrogen (N) atoms within the heterocycle. The Lewis acid B(C6F5)3 (BCF) was selected to coordinate with the N atoms in the polymer backbone. The number-averaged degree of polymerization (DP) estimated from gel permeation chromatography (GPC) was found to be 16 and 14, respectively, for P1 and P2, indicating that the average number of coordination sites is 32 and 28, respectively, per chain of P1 and P2. Titration experiments conducted by gradually adding BCF to the polymer solutions revealed red-shifted absorption bands. The terminal of the titration was found upon the addition of 40 eq. and 32 eq. of BCF to P1 and P2 solutions, respectively. By adding excess BCF to fully coordinate with the N atoms, the optical bandgaps declined from 2.10 eV (P1) to 1.49 eV (P1/BCF) and from 2.08 eV (P2) to 1.32 eV (P2/BCF). Meanwhile, the LUMO levels estimated from electrochemistry experiments were also depressed from 3.60 eV (P1) to 3.96 eV (P1/BCF) and from 3.59 eV (P2) to 4.12 eV (P2/BCF). Electrostatic potential surface (EPS) maps deduced from Gaussian simulations revealed that electron density located on the polymer backbone was withdrawn by BCF, transforming the polymer skeleton from electron rich to electron deficient. This revealed the reason for the decrease in the bandgaps and the reduction of the energy levels upon coordination with BCF.


 Please wait while we load your content...
Please wait while we load your content...