Histidine-assisted synthesis of CeO2 nanoparticles for improving the catalytic performance of Pt-based catalysts in methanol electrooxidation†
Abstract
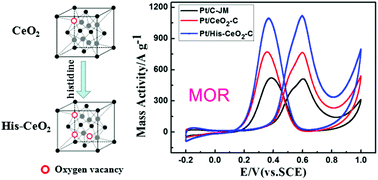
His-CeO2 nanoparticles were synthesized under urea hydrolysis conditions using histidine as the growth-directing agent. Characterization by transmission electron microscopy, powder X-ray diffraction spectroscopy, photoluminescence spectroscopy and X-ray photoelectron spectroscopy, indicated that the His-CeO2 nanoparticles, which had a particle size of 12.2 nm, possessed a higher content of Ce3+ species and oxygen vacancies than bare CeO2 nanoparticles. Pt/His-CeO2-C electrocatalysts were constructed by using His-CeO2 nanoparticles as co-catalysts and exhibited enhanced catalytic performance in the methanol oxidation reaction. Because His-CeO2 co-catalysts supply sufficient OHads sites and show strong interactions with Pt species, Pt/His-CeO2-C had better antipoisoning properties than Pt/CeO2-C. In cyclic voltammetry tests, the mass activity for Pt/His-CeO2-C was up to 1116.9 A g−1, which is approximately 2.9 and 2.2 times greater than that of Pt/C-Z and commercial Pt/C-JM, respectively. These results pave the way for manufacturing Pt-based electrocatalysts for methanol electrooxidation with biomolecule-mediated modulation of the synergistic effects between the Pt species and metal oxides.


 Please wait while we load your content...
Please wait while we load your content...