Synthesis and proton conductivity of two novel molybdate polymers†
Abstract
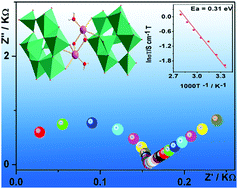
Two novel molybdate polymers, H4[Co(phen)3]2[NaO(H2O)(τ-Mo8O26)]2·2H2O (1) and H4[TEDA] [γ-Mo8O26]·3H2O (2), (phen = 1,10′-phenanthroline, TEDA = triethylene diamine), were successfully synthesized by a hydrothermal method with MoCl5. Single-crystal X-ray diffraction shows that 1 was a dimer-octamolybdate polymer connected by two Na+ cation, and it is the first molybdate dimer-polymer of [τ-Mo8O26]. Compound 2 was an extended 1D octamolybdate chain connected by contiguous [γ-Mo8O26]. Both 1 and 2 have 1D hydrogen-bond chains for proton-conducting pathways and free H+ counter cations as proton donors. These features make them suitable for potential proton-conducting solid crystal materials. The outstanding conductivities of 3.06 × 10−3 S cm−1 for 1 and 4.18 × 10−4 S cm−1 for 2 were achieved at 85 °C under 97% RH.


 Please wait while we load your content...
Please wait while we load your content...