Two isomers of a bis(diphenylphosphino)phosphinine, and the synthesis and reactivity of Ru arene/Cp* phosphinophosphinine complexes†‡
Abstract
The reaction of 4,6-di(tert-butyl)-1,3,2-diazaphosphinine (3) with two equivalents of MeC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CPPh2 gave two isomeric products, 2,6-bis(diphenylphosphino)-3,5-dimethylphosphinine (5) and 2,5-bis(diphenylphosphino)-3,6-dimethylphosphinine (6), which were successfully separated and their molecular structures determined by X-ray crystallography. Although the 2,6-bis(iminophosphorano)phosphinine 7 was readily synthesised from 5 using mesityl azide, its coordination to late transition metals was not achieved. The reaction of 2-diphenylphosphino-3-methyl-6-trimethylsilylphosphinine (1) with [{Ru(Cl)(μ-Cl)(p-cymene)}2] generated two products: cis-[Ru(Cl)2(1)2] (2) and the dinuclear species [Ru(μ-Cl)3(p-cymene)Ru(Cl)(1)] (8), which was characterised by single crystal X-ray diffraction. The reaction of 1 with [{Ru(Cl)(μ-Cl)(C6Me6)}2]/NH4PF6 led to cleavage of the SiMe3 group and addition of H2O across a P
CPPh2 gave two isomeric products, 2,6-bis(diphenylphosphino)-3,5-dimethylphosphinine (5) and 2,5-bis(diphenylphosphino)-3,6-dimethylphosphinine (6), which were successfully separated and their molecular structures determined by X-ray crystallography. Although the 2,6-bis(iminophosphorano)phosphinine 7 was readily synthesised from 5 using mesityl azide, its coordination to late transition metals was not achieved. The reaction of 2-diphenylphosphino-3-methyl-6-trimethylsilylphosphinine (1) with [{Ru(Cl)(μ-Cl)(p-cymene)}2] generated two products: cis-[Ru(Cl)2(1)2] (2) and the dinuclear species [Ru(μ-Cl)3(p-cymene)Ru(Cl)(1)] (8), which was characterised by single crystal X-ray diffraction. The reaction of 1 with [{Ru(Cl)(μ-Cl)(C6Me6)}2]/NH4PF6 led to cleavage of the SiMe3 group and addition of H2O across a P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to generate [Ru(C6Me6)(1-OH-2-PPh2-3-MePC5H4)][PF6] (9). The reaction of 1 with [{Ru(Cp*)(μ3-Cl)}4] yielded [Ru(Cp*)(Cl)(1)] (10) which readily reacted with H2O across a P
C bond to generate [Ru(C6Me6)(1-OH-2-PPh2-3-MePC5H4)][PF6] (9). The reaction of 1 with [{Ru(Cp*)(μ3-Cl)}4] yielded [Ru(Cp*)(Cl)(1)] (10) which readily reacted with H2O across a P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to form [Ru(Cp*)(Cl)(1-OH-2-PPh2-3-Me-6-SiMe3PC5H3)] (11). Neither 9, 10, 11 or cis-[Ru(Cl)2(dppm)2] were effective precatalysts for the transfer hydrogenation (TH) of acetophenone, unlike 2 which in addition was also found to catalyse the TH of benzophenone at 82 °C (0.1 mol% 2 with 0.5 mol% KOtBu in iPrOH), with much lower activity for 2-fluorobenzaldehyde and 4-methylcyclohexanone. 11 was a competent precatalyst for the hydrogen-borrowing upgrading of EtOH/MeOH to isobutanol, albeit in lower yields compared to 2.
C bond to form [Ru(Cp*)(Cl)(1-OH-2-PPh2-3-Me-6-SiMe3PC5H3)] (11). Neither 9, 10, 11 or cis-[Ru(Cl)2(dppm)2] were effective precatalysts for the transfer hydrogenation (TH) of acetophenone, unlike 2 which in addition was also found to catalyse the TH of benzophenone at 82 °C (0.1 mol% 2 with 0.5 mol% KOtBu in iPrOH), with much lower activity for 2-fluorobenzaldehyde and 4-methylcyclohexanone. 11 was a competent precatalyst for the hydrogen-borrowing upgrading of EtOH/MeOH to isobutanol, albeit in lower yields compared to 2.
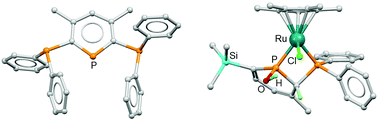


 Please wait while we load your content...
Please wait while we load your content...